 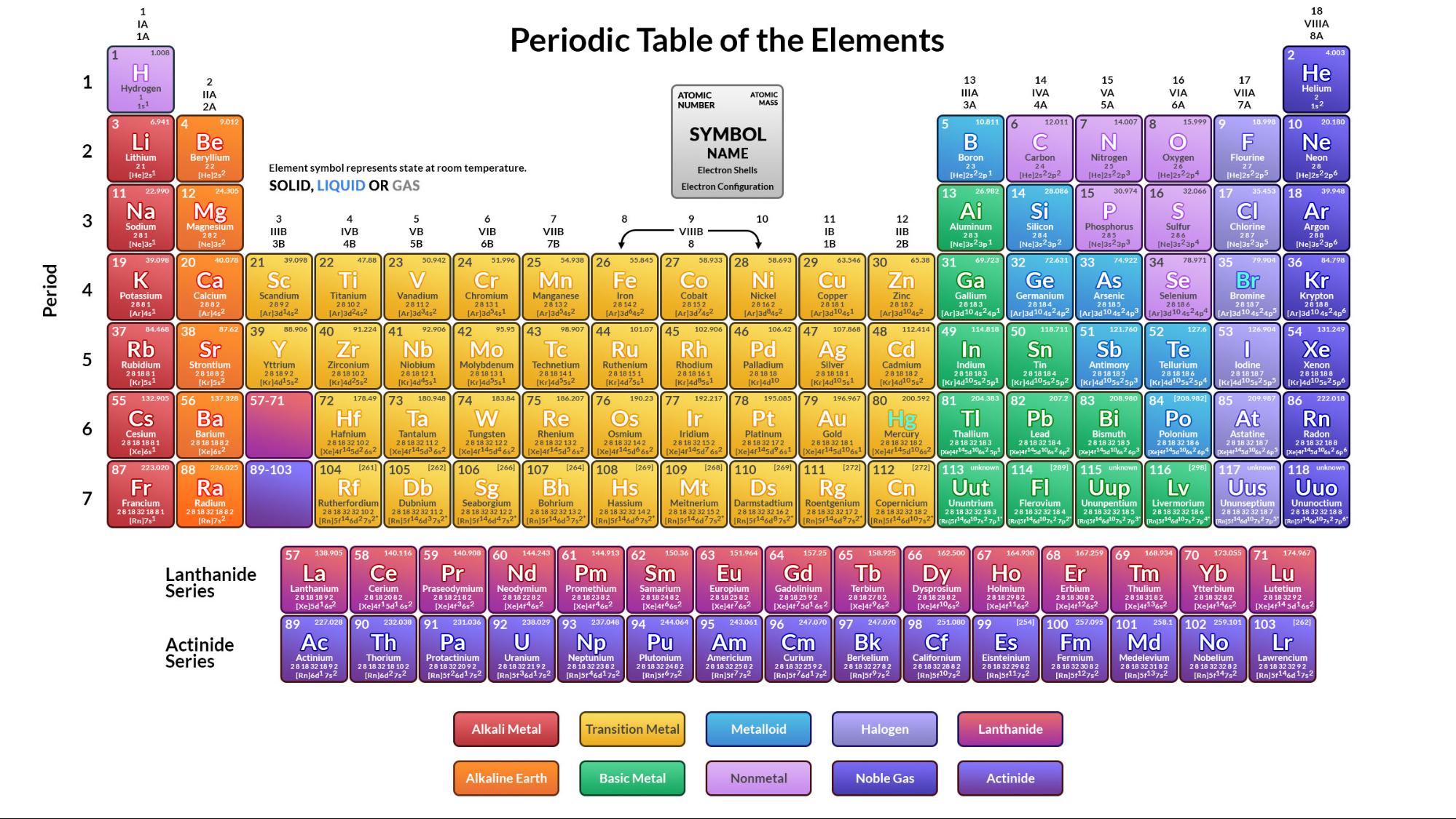
It's thought that another one of these magic numbers lies just beyond the range of currently known elements, which means we could potentially discover some new elements that are relatively stable and extremely massive. There are believed to be certain numbers/ratios that are inherently more stable. Formulas indicate the type of compounds formed by each group, with R standing for any element and superscripts. The column headings Reihen and Gruppe are German for row and group. Hence, larger atoms become more unstable. 3: Mendeleev’s Periodic Table, as Published in the German Journal Annalen der Chemie und Pharmacie in 1872. The ratio of neutrons to protons gradually increases to try to compensate, but this results in the neutrons getting more unstable themselves, resulting in more opportunity for weak forces to destabilize them. 1: Periodic table by Dmitri Mendeleev, 1871. After the development of quantum mechanics, it was shown that the halogens all had seven valence electrons, supporting their original placement into the same group on Mendeleevs periodic table. In 1869, Russian chemist Dmitri Mendeleev created the framework that became the modern periodic table, leaving gaps for elements that were yet to be discovered. The particles that comprise atomic nuclei are termed nucleons. Atomic nuclei are not elementary particles, they are themselves composed of more fundamental particles. In comparison to an atom, it is much more smaller and contains most of the mass of the atom. With all of that to consider, as you get larger and larger nuclei with more protons, the electromagnetic forces increase, as well as the distances in the nuclei, putting more strain on the nuclear forces holding it all together. As a gas or vapor, the halogens all had a pungent odor. atom: The smallest possible amount of matter which still retains its identity as a chemical element, consisting of a nucleus surrounded by electrons. In physics, the atomic nucleus is the central part of an atom. The term effective nuclear charge should be used in place of nuclear charge because effective nuclear charge takes into account the behaviour of all electrons whether they constitute the valence shell or are present in the inner shell. Neutrons will help add to this nuclear force, but they tend to become unstable when they get into too great of concentration and the weak force will cause them to decay into protons. The nuclear charge is the total charge in the nucleus of all the protons. As its name implies, this particle carries no electrical charge. The other nuclear particle is the neutron. Fusion allows these protons to get close enough together for the stronger, yet short ranged, nuclear force to overpower the electromagnetic force. This means that in any electrically neutral atom, the number of protons in the nucleus (often referred to as the nuclear charge) is balanced by the same number of electrons outside the nucleus. The electromagnetic force of the protons wants to push them away from each other. (CC-BY-NC-SA Kathryn Haas) Where F is the force, k is Coulombs constant, q 1 and q 2 are the charges on the two particles, and r is the distance between the particles. Coulombs law applied to the attractive force between a nucleus and an electron. Negative and positive charges of equal magnitude cancel each other out.There are several forces at work inside the nuclei of atoms. Coulombs law is defined as: (1.1.1.1) F k q 1 q 1 r 2. \)) are useful, because, as you can see, the mass of a proton and the mass of a neutron are almost exactly \(1\) in this unit system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
